|
4/2/2024 0 Comments Dalton's atomic theory model
Most of the mass of an atom was concentrated in an extremely small volume. 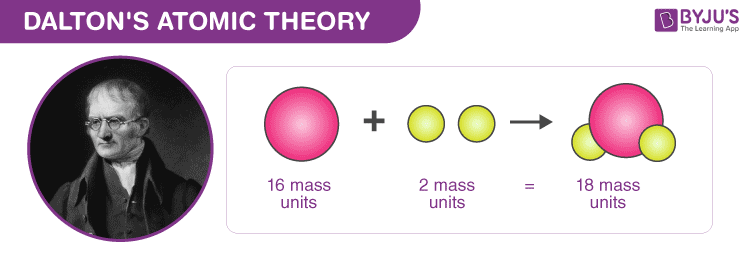
In the early 1900s, Thomson discovered that atoms had a nucleus held. In the year 1903, Ernest Rutherford conducted an experiment in which he bombarded a thin sheet of gold with α-particles and then studied the trajectory of these particles after their interaction with the gold foil. Through this experiment, Rutherford determined that the particles must have been scattered in all directions with very few exceptions and discovered that atoms are mostly empty spaces. This was called the Rutherford Atomic Model. Modern atomic theory, though evolved in two centuries, holds much of Dalton’s atomic theory.Įrnest Rutherford was a British scientist who conducted an experiment and proposed the atomic structure of different elements. Later, JJ Thompson discovered electrons and Rutherford worked on the model to discover the nucleus.įinally, Niels Bohr’s model and the Quantum mechanical model provided the modern atomic model as it is known today. The Atomic theory got modified with the contribution of many after Dalton, namely, Chadwick, JJ Thompson, Ernest Rutherford, Niels Bohr etc. Dalton’s model was revolutionary for the period and gave much to the new chemists to research upon. It can not prove the difference in properties of charcoal, diamond, and graphite allotropes of carbon.ĭalton’s atomic theory contributed a lot to modern atomic theory. The theory fails to explain the difference in properties of allotropes. Atoms of different elements when having the same mass, are called isobars. This fails to explain the formation of complex organic compounds.ĭalton’s model failed to explain the existence of isobars. These are called isotopes.Īccording to the theory of Dalton, compounds are formed when atoms combine in whole numbers. However, a single element having different atomic masses has been observed. Subatomic particles like protons, electrons, and neutrons have been discovered since then.ĭalton’s atomic theory states atoms of an element are identical in mass. In further research, his theories were proven wrong.Īs it was found, later on, atoms are not indivisible. The number of atoms before and after a chemical reaction remains the same.Īlthough Dalton’s atomic theory marked a significant turning point in the research regarding the matter, the theory wasn’t entirely faultless. Different types of atoms are joined together to form compounds.Ī chemical reaction is a rearrangement of atoms, where the formation of new products occurs due to the rearrangement of atoms in the reactant. masses, sizes, shapes, and other chemical properties.Ītoms can neither be created nor be destroyed or subdivided into a chemical reaction.Ītoms of different elements combine in whole numbers in a simple but fixed ratio to form compounds. Different elements have different properties i.e. So, they have the same size, shape, mass, and chemical properties. Based on these laws Dalton’s atomic theory states the following postulates:Ītoms are considered to be a matter which is made up of very small, indivisible particles.Īll the atoms in an element are identical. The law of conservation of mass and constant proportions are the basis that helps explain Dalton’s atomic theory. The definition of dalton's atomic theory brought the novel concept of calculating relative atomic weights. Although connections of his work have been made with several other chemists of the time. Dalton’s idea for the theory is believed to be inspired by the physical properties of gases. The theory of Dalton was published in the paper “New Chemical Philosophy”. However, the ideas from researches of methane and ethylene might have helped define Dalton’s atomic theory at the time. Dalton’s model came almost two millennia later and brought further light to the topic. Democritus was known to be the first to suggest that matter is made up of particles. The matter has been a subject of fascination since the beginning. (Image to be added soon) Dalton’s Atomic Model 
Each atom was circular and bears different symbols. 
Dalton’s model suggested the atom to be a ball-like structure that cannot be further divided. He considered the atom as the smallest, indivisible unit of matter and wrote several postulates. Dalton's atomic model showed the way to many future works, researches regarding atomic theory, even though his conclusions were rather incorrect. He was a British physicist, chemist, and meteorologist who is well known for many of his contributions to the pioneering research of atoms, the law of partial pressures, Daltonism, etc. \)) More important, by measuring the extent of the deflection of the cathode rays in magnetic or electric fields of various strengths, Thomson was able to calculate the mass-to-charge ratio of the particles.The introduction to the early theory of the atom was done by a scientist named John Dalton (1766-1844).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
